Imagine trying to recreate a world-class secret recipe, but you aren't allowed to see the ingredient list or the cooking instructions. You can only taste the final dish and guess what went into it. This is exactly what pharmaceutical companies face when developing complex generics. While making a standard generic tablet is relatively straightforward, replicating a drug that uses nanotechnology, a specialized inhaler, or a slow-release injectable is a scientific mountain climb.
The core of the struggle is proving Bioequivalence is the absence of a significant difference in the rate and extent to which the active ingredient becomes available at the site of drug action. For a basic pill, you just measure how much drug ends up in the blood. But for a complex generic, the "site of action" might be deep in the lungs or embedded in the skin, where taking a blood sample tells you almost nothing about whether the drug is actually working.
The Five Faces of Complexity
Not all generics are created equal. The FDA generally splits these challenging products into five buckets based on what makes them difficult to replicate:
- Complex Active Ingredients: These involve Peptides, polymers, or naturally sourced ingredients that don't behave as predictably as simple chemical molecules.
- Complex Formulations: This includes Liposomes and colloids-essentially tiny fat bubbles or particles that carry the drug.
- Complex Delivery Routes: Think of gels, emulsions, or suspensions used in the eyes, ears, or on the skin.
- Complex Dosage Forms: These are the "long-haul" drugs, like extended-release injectables or metered dose inhalers.
- Drug-Device Combinations: Products where the hardware (like a specialized pump or inhaler) is just as important as the medicine inside.
Because of these hurdles, the success rate is sobering. While over 80% of traditional small-molecule generics get approved, only about 10-15% of complex generic applications make the cut. It's a high-stakes game of scientific precision.
Why Blood Tests Aren't Enough
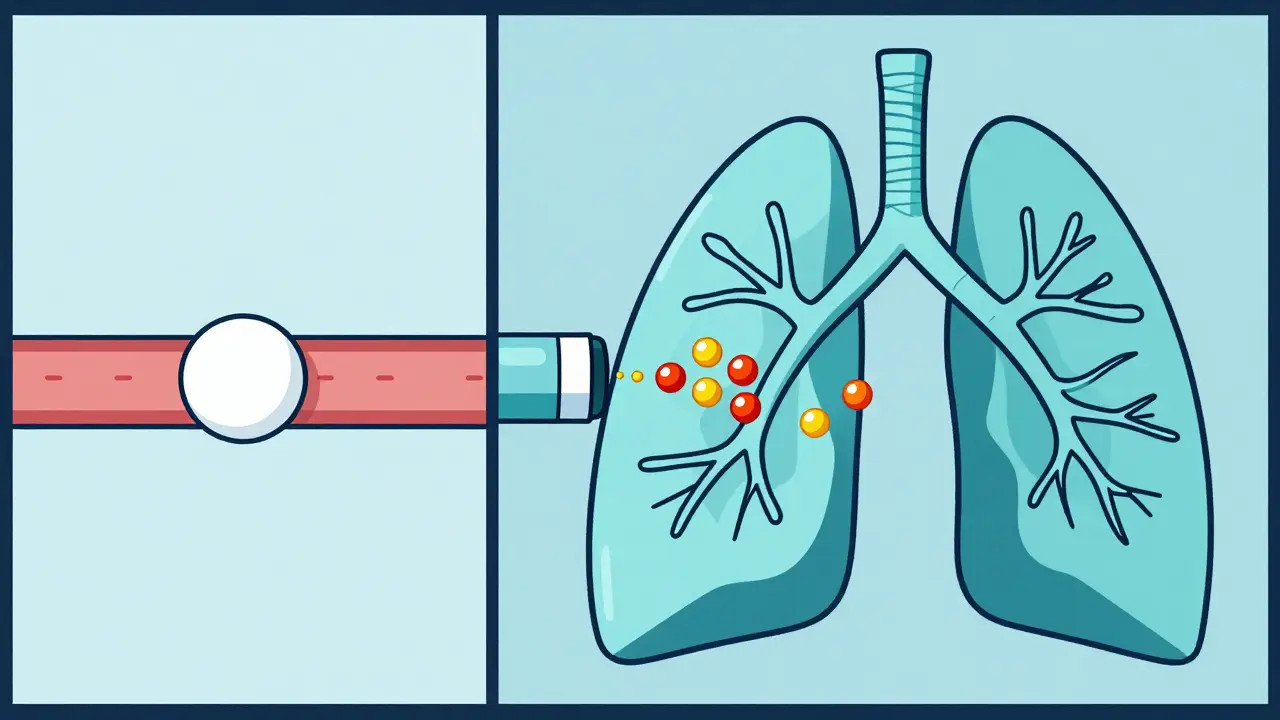
In a standard bioequivalence (BE) study, researchers look for the 90% confidence interval of the AUC (area under the curve) and Cmax (maximum concentration) to fall between 80% and 125%. If the generic drug hits those numbers, it's usually a win. But for a Topical Dermatological Product, the drug is meant to treat a rash on the surface of the skin, not enter the bloodstream. Measuring blood levels becomes irrelevant.
This leaves manufacturers in a bind. How do you prove a cream is penetrating the skin layers exactly like the brand-name version? You can't easily biopsy every patient's skin. Similarly, for inhaled medications, the challenge is "lung deposition." If the particle size is slightly off-say, larger than 10 micrometers-the drug hits the back of the throat instead of reaching the deep alveoli of the lungs. This tiny physical difference can make a generic drug a total failure in the eyes of regulators.
| Feature | Traditional Generics | Complex Generics |
|---|---|---|
| Primary BE Metric | Plasma concentration (Blood) | Local site action / Product performance |
| Development Time | Standardized timeline | 18-24 months additional time |
| Approval Rate | High (>80%) | Low (10-15%) |
| Main Risk | Chemical purity | Manufacturing & stability variance |
The "Reverse Engineering" Nightmare
Generic developers don't get a blueprint of the Reference Listed Drug (RLD). They have to engage in "de-formulation." This is a painstaking process of reverse-engineering the brand-name product to identify its Critical Quality Attributes (CQAs).
One tiny change in an inactive ingredient-an excipient-can trigger a disaster. For instance, a different salt form of the active pharmaceutical ingredient (API) might change how a drug dissolves or how stable it remains over six months. Stability is a huge headache; oxidation and hydrolysis can degrade a complex formulation long before the expiration date, meaning the drug might be bioequivalent on day one but useless by day ninety.
Moreover, global companies face a fragmented regulatory map. The European Medicines Agency (EMA) might demand a different proof of equivalence than the FDA. This forces companies to run duplicative, expensive tests, adding millions to the development cost.

Modern Solutions: Moving Beyond the Lab
To bridge the gap, the industry is moving toward Quality by Design (QbD). Instead of making a batch and hoping it works, manufacturers now plan for stability and BE from the first day. This means running rigorous compatibility programs between the API and excipients before a single prototype is ever built.
We're also seeing a shift toward Physiologically-based Pharmacokinetic (PBPK) modeling. These are sophisticated computer simulations that model how a drug moves through the body. In some cases, PBPK modeling can reduce the need for actual human BE studies by 40-60% by proving the mechanistic link between the drug's chemistry and its clinical outcome.
The FDA has also stepped up by creating the Complex Generic Drug Product Development Program. By hosting workshops and publishing specific guidance documents for things like testosterone topical products or budesonide inhalers, they are trying to lower the barrier to entry. The data shows it works: companies that engage with the FDA early through these programs see about 35% higher approval rates.
Looking Ahead: The Billion-Dollar Opportunity
Despite the headaches, the incentive is massive. There's a roughly $120 billion opportunity in the US alone, with hundreds of complex drugs still lacking generic alternatives. As the need for cheaper specialty meds grows, we can expect to see more advanced imaging for skin penetration and better in vitro lung models.
The industry is moving toward a future where we don't just guess the "secret recipe," but use high-tech tools to map the molecular behavior of drugs in real-time. It's no longer just about copying a pill; it's about mastering the physics of how medicine enters the human body.
Why are complex generics more expensive to develop than regular ones?
They require significantly more resources-often 2.5 to 3 times more than traditional generics. This is due to the need for "de-formulation" (reverse engineering), specialized equipment for particle size analysis, and longer stability testing periods to ensure the drug doesn't degrade.
What is a "local site of action" and why does it matter for BE?
A local site of action is where a drug works directly, such as the surface of the eye or the lining of the lungs, rather than absorbing into the blood to travel throughout the body. Because the drug doesn't need to enter the bloodstream to work, traditional blood-concentration tests cannot prove if the generic is delivering the medication effectively to that specific spot.
Can PBPK modeling replace human clinical trials?
It doesn't always replace them entirely, but it can significantly reduce the requirements. By using mathematical models to simulate drug absorption and distribution, researchers can bridge gaps where traditional studies are impractical, potentially reducing study requirements by 40-60% for certain products.
What happens if a complex generic fails a bioequivalence test?
Failure rates at the BE stage for complex generics often exceed 70%. If a product fails, the manufacturer must either reformulate the drug (change ingredients or the manufacturing process) or provide alternative scientific data to prove the drug is still safe and effective, which can add years to the development timeline.
How does the FDA help companies make complex generics?
The FDA provides guidance documents, hosts scientific workshops, and offers the Complex Generic Drug Product Development Program. These resources help companies align their development pathways with regulatory expectations early on, which historically increases approval rates by about 35%.

Hudson Nascimento Santos
It's wild how much of our health depends on the physics of a particle. We talk about medicine as chemistry, but this really shows it's just as much about engineering. If the delivery mechanism fails, the most potent drug in the world is just expensive waste.