Pharmaceutical production — how your medicine gets made and why it matters
Ever wondered what happens between a lab recipe and the pill bottle on your shelf? Pharmaceutical production covers everything from raw ingredients and active pharmaceutical ingredients (APIs) to mixing, tableting, quality checks, and packaging. Knowing a bit about that process helps you spot good products, avoid fakes, and understand why prices and safety vary.
At the factory level, a few steps matter most: sourcing APIs, following Good Manufacturing Practices (GMP), precise formulation, stability testing, and proper packaging. GMP is the baseline — it means the plant keeps clean records, controls contamination, and routinely tests batches. If a manufacturer has FDA, EMA, or local regulator approvals noted on their site, that’s a solid sign they follow recognized standards.
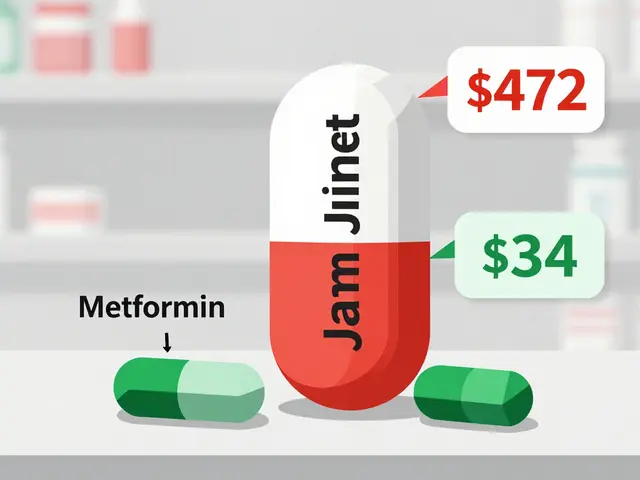
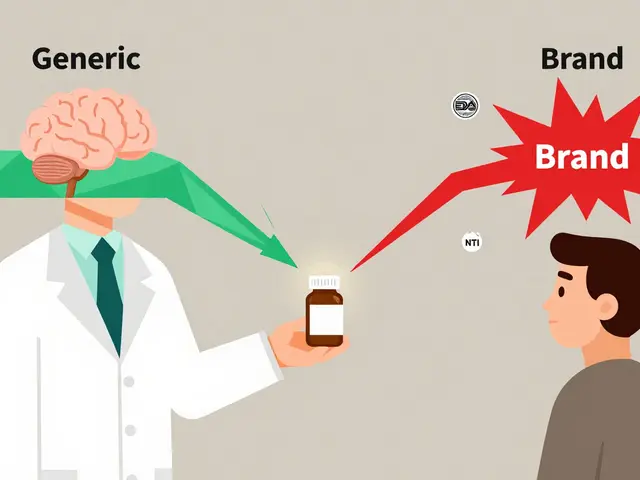
How does production affect you? First, quality and safety. A well-made batch behaves the same each time — same strength, same shelf life. Second, cost. Generic drugs save money because manufacturers use the same approved API and make it more cheaply once patents expire. But cheaper doesn’t always mean low quality: a licensed generic made in a GMP facility is usually safe and effective.
Spotting red flags is simple if you know what to look for. Watch out for packages missing batch numbers or expiry dates, misspelled brand names, or flimsy seals. Prices that are dramatically lower than usual can mean counterfeit or diverted meds. On the other hand, clear labeling, a batch/lot number, and a manufacturer address are positive signs.
Buying medicine online — quick safety tips
Shopping for meds online? Ask for a prescription, confirm the pharmacy is licensed, and check for a physical address and phone number. Search your country’s pharmacy board to confirm licenses. If a site refuses a prescription or pressures you to buy without one, move on. Also look for third-party pharmacy verification seals and read recent user reviews — not perfect, but helpful.
Production quality intersects with supply chain issues, too. APIs can come from many countries; interruptions or poor raw-material control can change drug availability and price. That’s why some articles here dig into manufacturing trends, company reviews, and comparisons between branded and generic options — practical stuff that affects what you can actually buy.
Quick checklist before you take a chance
- Check for GMP or regulator approvals on the manufacturer’s page.
- Verify pharmacy licensing with local boards.
- Look for batch numbers, expiry dates, and tamper seals.
- Get and keep a prescription for prescription-only meds.
- Avoid extreme discounts and high-pressure sales tactics.
This tag collects articles that explain production issues, show how to buy specific medicines safely, compare alternatives, and review online pharmacies. Browse the posts to learn practical steps for protecting your health and your wallet — and reach out if you want a plain answer about a specific drug or supplier.
Doxepin is a common antidepressant, but making it isn't as clean as many people think. The process can leave behind chemical waste, pollute water, and contribute to greenhouse gas emissions. Understanding the impacts helps people make smarter choices about medication use and disposal. This article breaks down where the problems show up and what can be done to lower the footprint. There's more going on behind the scenes of that little pill than most imagine.
Continue reading...